Pool Water Chemistry

Points to consider:
The information given in this module follows guidelines issued by Australian Standards, Queensland. Government Health Water quality guidelines for public aquatic facilities and general industry parameters. This will always be Billabong's policy.There is no definitive set of water chemistry for all pools. Every pool is a separate entity. There is a marked difference in chemical demand for a domestic pool and a commercial pool. We will in most cases be discussing commercial pools, domestic requirements are less demanding but generally allied
Queensland Health's water chemistry protocols are crucial for maintaining water quality and safety in the region. They provide guidelines for monitoring and managing water sources to safeguard public health, the environment, and promote sustainable water practices.
Water quality guidelines for public aquatic facilities, Queensland Health.
Training Topics
Exploring the essential topics of swimming pool chemistry is vital. Proper water balance and knowledge of the key chemical components are imperative to maintain a clean, safe, and enjoyable pool.
pH
Exploring the significance of pH levels in swimming pools is crucial for maintaining optimal water quality. It's essential to understand how to keep these levels balanced to ensure a safe and enjoyable swimming experience.
Cyanuric Acid (stabiliser)
Cyanuric acid (CYA), commonly referred to as a chlorine stabilizer or pool conditioner, is vital for the upkeep of swimming pools. It's important to understand its significance and learn the best practices for its management.

Testing
Testing your pool water is essential to ensure water quality. Accurate testing results in a clean and safe swimming environment.
Overview of a Pool
Let’s explore, an overview of a swimming pool.
Total Alkalinity
Exploring the significance of Total Alkalinity (TA) in swimming pool chemistry reveals its crucial role in preserving water balance.
Totally Dissolved Solids
Total Dissolved Solids (TDS) are crucial for maintaining the quality of swimming pool water. Let's explore the concept of TDS, understand its importance, and learn how to control it effectively.

Chemical Parameters
Proper swimming pool water chemistry maintenance is crucial for ensuring water quality and swimmer comfort. We will delve into the key chemical parameters.
Halogens, Chlorine, Sanitisers
Exploring the role of halogens as disinfectants reveals that this group of chemical elements, which includes chlorine, fluorine, bromine, and iodine, possesses distinctive properties that render them effective in disinfection applications.
Calcium Hardness
Exploring the significance of calcium hardness in swimming pool chemistry is crucial. Calcium hardness denotes the measure of dissolved calcium present in the water of your pool. Understanding its importance is key.
L.S.I. Saturation Index
The Langelier Saturation Index (LSI), or swimming pool saturation index, is a vital element of pool water chemistry. It's important to understand its significance and the methods to maintain it, ensuring a safe and healthy swimming environment.

Pool Turnover
Swimming pool turnover is essential for preserving water quality and guaranteeing a safe, enjoyable swimming experience. Let's delve into its significance.
Training Topics
OVERVIEW OF A POOL
THE STRUCTURE
BASIC AIMS FOR PRESENTATION
THE VOLUME
TURN OVER TIME
CHLORINE
THE PRODUCT
FREE TOTAL & COMBINED
HYPOCHLOROUS ACID HYPOCHLORITE
ION RESIDUAL
CHLORINE CHLORAMINES BREAKPOINT
CHLORINATION TYPES OF CHLORINE
CHLORINE VS SALT CHLORINATION
pH
THE SCIENTIFIC DEFINITION
THE PRACTICAL DEFINITION
A pH SUITABLE FOR POOLS
THE pH OF PRODUCTS USED IN POOLS
TOTAL ALKALINITY
THE NEED
THE ADJUSTERS
THE EFFECTS
ON pH
ON WATER QUALITY
ON EQUIPMENT
CALCIUM HARDNESS
THE NEED
THE PRODUCT
THE EFFECTS
ON WATER QUALITY
ON EQUIPMENT
ON SURFACES
ON SALT CHLORINATORS
LANGELIER INDEX
THE NEED
THE COMPONENTS
CALCULATING THE INDEX
INTERPRETATION OF THE INDEX
CYANURIC ACID
THE NEED
THE EFFECT ON CHLORINE ACTIVITY
APPLICATION FOR POOLS AND SPAS
CHLORINE BLOCK
FREQUENCY OF TESTING
COMMERCIAL POOLS
DOMESTIC POOLS
LOG BOOKS
MICROBIOLOGICAL
TESTING
FOR PLATE COUNT
FOR PSEUDOMONAS AND
AERUGINOSA
FOR E. COLI
(TEST KITS)
PHOTOMETER
COMPARATOR
TITRATION
BALANCE CHARTS
CHEMICAL PARAMETERS
THE IMPORTANCE OF
BALANCED WATER CHEMISTRY
CURRENT CHEMICAL
PARAMETERS
POOL TURNOVER
DISCUSSING THE IMPORTANCE OF POOL TURNOVER.
The Overview of a Swimming Pool
Types of Swimming Pools:
In-Ground Pools:
These are permanent pools built into the ground. They can be made of various materials, including vinyl, fibreglass, or concrete.
Vinyl Pools (above ground)
The least expensive option. A frame is erected onto an excavation, and a heavy-duty vinyl liner is installed.
Fibreglass Pools:
Pre-moulded in various shapes and sizes, with steps and benches already in place.
Concrete Pools:
Also known as wet shotcrete or Gunite pools. A plaster finish is applied over the concrete surface.
Pool Shapes:
Lap Pools:
Narrow and ideal for training or exercise. Some are as long as Olympic pools (25 meters).
Recreational Pools:
Typically rectangular, with one end shallow and the other deep enough for diving (9 to 11 ft.). They come in various sizes.Free-form Pools: Organic shapes that blend well into the backyard landscape.
Pool Construction Methods:
Vinyl, fibreglass, and concrete are common choices.
Vinyl pools are cost-effective, while fibreglass pools come pre-moulded.
Concrete pools (shotcrete or Gunite) offer durability and customisation.
Remember, choosing the right pool involves considering your needs, budget, available space, and design preferences.
What maintenance is required for a swimming pool?
Please refer below for the Billabong procedure on how to treat new concrete pool surfaces and fibreglass pool surfaces.
Regular maintenance is essential to keep your swimming pool clean, safe, and enjoyable. Here are the key tasks for maintaining your pool:
Pool Water:
Keep the water clean, clear, and balanced.
Test and adjust pH levels (7.4 – 7.6).
Maintain free available chlorine (1.0 – 3.0 ppm).
Top up the water level as needed.
Pool Interior:
Regularly clean the pool walls, floor, corners, and ladders to prevent algae and debris buildup.
Address any algae or mould promptly.
Pool Filter System:
Ensure your filter system (cartridge, sand, or *D.E., * which are not in common use) are functioning properly.
Regularly clean or backwash the filter to remove dirt and contaminants.
Skimmers and Returns:
Skimmers pull water into the filter for cleaning. Returns distribute the cleaned water back into the pool. Remember, consistent maintenance ensures a healthy and inviting swimming experience!
HALOGENS

This group of elements, known as the **halogens**
(group 17 of the periodic table), is full of surprises and noteworthy characteristics that make it a fascinating subject of study:
1. **Reactivity and Electron Affinity**
Each halogen has seven electrons in its outer shell, making them highly reactive as they eagerly seek one more electron to achieve a stable, noble gas configuration. This characteristic drives a wide range of chemical reactions, such as forming the salts we use daily—for example, when chlorine combines with sodium, it creates table salt.
2. **Distinct Physical States Across the Group**
There’s an intriguing trend in the halogen family: as you move down the group, the elements progress from gases to liquids to solids at room temperature. Fluorine and chlorine are gases, bromine is a liquid (and is notably the only non-metallic element liquid at room temperature), and iodine usually appears as a solid with a shiny, lustrous appearance. Astatine, being far down the group, is also a solid, though it’s extremely rare and radioactive.
3. **Key Applications and Impact** :
**Fluorine**is central to modern chemistry. Its high electronegativity not only makes it aggressively reactive but also useful in creating strong, durable compounds like Teflon or in dental products such as toothpaste.
**Chlorine** is indispensable in water treatment and sanitation. Despite its benefits, it’s also had a notorious past—used as a chemical warfare agent during World War I.
**Bromine** finds its niche in flame retardants and certain pharmaceuticals, while
**Iodine** is critical for human health, particularly for thyroid function, and is widely used as an antiseptic in medical settings.
**Astatine**, on the other hand, is so scarce and radioactive that our understanding of it is mostly theoretical, with only minute amounts occurring naturally.
4. **Chemical Bonds and Environmental Roles**
Halogens tend to form diatomic molecules (like F₂ and Cl₂), and their reactions with metals yield a bulk of our familiar salts. Beyond their everyday roles, halogens have environmental implications; chlorine compounds, for example, are central in discussions about ozone layer depletion and the broader impacts of industrial chemicals on our atmosphere.
These facts hint at just a fraction of the halogens' role, from everyday applications to profound chemical properties. If you’re curious about how their reactivities compare or want to dive into how halogens influence environmental chemistry, there’s a wealth of fascinating details to explore. The aspects of the halogen family interests us the most relate to swimming pool sanitation.

CHLORINE DISSOCIATION

CHLORINE ADDED TO WATER.
PH of water 7.6
+ pH
39.0 % --------------> 61.0%
HOCL <-------------- (H+) + (OCL-)
- pH
Hypochlorous Acid Hydrogen Ion Hypochlorite Ion
Equilibrium Reaction:
The dissociation of HOCl can be represented as an equilibrium reaction:
HOCl⇌H⁺+OCl⁻
The balance between HOCl and OCl⁻
depends on the pH level:
At lower pH (more acidic conditions), there’s more HOCl (active form).As pH increases, the proportion of OCl⁻
(less active form) rises TEST KIT DPD NO1 OR R0001 / R0002 free chlorine (FC) READING OF 2.0 ppm
CHLORINE RESIDUAL
Chlorine residual refers to the amount of chlorine remaining in your pool water after it has reacted with organic matter, such as sweat, urine, and other contaminants. This residual chlorine is crucial for keeping your pool clean and free of harmful bacteria, viruses, and algae.
There are three main types of chlorine to understand:
Free Chlorine: This is the amount of chlorine available to sanitize your pool. It’s effective but can be unstable.
Combined Chlorine: Also known as chloramines, this is chlorine that has already reacted with contaminants. It’s less effective and can cause that characteristic chlorine smell.
Total Chlorine: This is the sum of free chlorine and combined chlorine in your pool.
Maintaining the right levels of chlorine residual is essential for effective pool sanitation. The ideal range for free chlorine in swimming pools is typically between 2.0 to 4.0 ppm (parts per million).
Effects of Temperature on Free Chlorine
Temperature has a significant impact on the effectiveness and stability of free chlorine in swimming pools. Here are the key points to consider:
Increased Chlorine Demand: Warmer Water: As the temperature rises, the demand for chlorine increases. This is because higher temperatures promote the growth of bacteria and algae, which chlorine needs to kill.
Rule of Thumb:
For every 12°C increase above 27°C, the amount of chlorine needed to maintain adequate levels can double.
Faster Chlorine Degradation:
Chemical Reactions: Higher temperatures accelerate chemical reactions, including the breakdown of chlorine. This means chlorine will work faster but also get used up more quickly.
Liquid Chlorine vs Granular Chlorine vs Salt Chlorination
Swimming pool chlorination is essential for maintaining clean and safe water. Here are the most common forms of swimming pool chlorination:
-
Liquid Chlorine (Sodium Hypochlorite):
-
This is a fast-acting form of chlorine that is added directly to the pool water. It’s effective for immediate sanitation and is often used for shock treatments.
-
-
Chlorine Tablets (Trichloroisocyanuric Acid Stabilized) & (Calcium Hypochlorite unstabilized):
-
These tablets dissolve slowly in the water and are typically placed in a floating dispenser or skimmer. They provide a steady release of chlorine, making them convenient for regular maintenance.
-
-
Granular Chlorine:
-
Similar to tablets, granular chlorine Calcium Hypochlorite unstabilized (Cal Hypo), Trichloroisocyanuric Acid Stabilized (Trichlor), Dichloro-S-Triazinetrione stabilized (Dichlor) can be added directly to the pool. It’s often used for shock treatments and can dissolve quickly in water.
-
-
Saltwater Chlorination:
-
This system uses a salt cell to convert salt into chlorine through a process called electrolysis. It provides a more stable chlorine level and is often preferred for its lower maintenance and softer water feel.
-
-
Stabilized Chlorine:
-
This type of chlorine contains cyanuric acid, which helps protect chlorine from being broken down by sunlight. It’s particularly useful for outdoor pools.
-
Each method has its pros and cons, depending on factors like pool size, usage, and personal preference.
Here’s a concise comparison of the chlorine content (by weight) in various disinfectant forms commonly used in water treatment and pool chemistry:
Chlorine Content by Compound
⚠️ Practical Considerations
-
Stabilized vs. Unstabilized: Trichlor and Dichlor contain cyanuric acid, which protects chlorine from UV degradation but can reduce effectiveness at high levels.
Trichlor adds less CYA per unit of chlorine than Dichlor, making it more efficient for long-term chlorination if CYA buildup is a concern.
-
Dichlor, especially the dihydrate form, introduces CYA more rapidly — useful if you're trying to raise stabilizer levels quickly, but risky for over-stabilization.
-
For every 10 ppm of free chlorine (FC) added:
-
Trichlor contributes about 6 ppm of CYA
-
Dichlor contributes about 9 ppm of CYA
This means Dichlor has a steeper CYA accumulation curve, which can lead to chlorine lock if not managed carefully — something you should be attuned to in your pool chemistry work.
-
Handling & Safety: Chlorine gas is potent but hazardous to store and handle. Liquid and solid forms are safer for residential use.
-
Application: Choice depends on system needs—e.g., pools, drinking water, industrial disinfection.

Chlorine vs. Cyanuric Acid Ratios

COMPARISON OF LIQUID AND SALT CHLORINATION
We are regularly asked "which is the better system to use, salt or chlorine?" The question itself is a contradiction in term and highlights the misconceptions surrounding salt chlorination systems. The following information has been produced purely for the purpose of comparison and draws no conclusions as to whether one system is superior to the other. This comparison only applies to domestic pools. Commercial pools have a much higher chlorine demand and fixed output over time dosing systems should not be used. Tablet and granular types of chlorine are not applicable to this comparison. The chlorine dosed system referred to in this comparison is specifically liquid chlorine (Sodium Hypochlorite), introduced by a chemical delivery pump. Points to consider when choosing a chlorination system are:-
1. Operating costs.
2. How manageable and efficient is the system.
3. Access to the pool equipment area for liquid chlorine and salt deliveries.
Liquid Chlorine: A peristaltic or diaphragm style pump is used to deliver the chlorine to the pool water. The electric dosing pump draws from a holding tank and injects a measured amount of chlorine over the daily run time of the pool pump. The pool pump and chlorine pump must run together, the quantity of chlorine dosed is regulated by a user set output control on the chlorine pump. The chlorine pump maximum output is specified to suit the pool pump calculated run time.
Liquid Chlorine is commercially manufactured at a minimum 10% strength, must be stored in a UV sound container and has a shelf life of 2 to 3 months.
Salt Chlorination:
A salt chlorinator produces chlorine by electrolysis. A salt pool operates with the same type and quantity of chlorine as that produced commercially (Sodium Hypochlorite). A salt pool is not a chlorine free pool. Salt in its own right does not sanitize pool water. A salt chlorination system can be broken down into three distinct components:-
1. The Power Pack.
2. The Cell.
3. The Electrolyte.
Power Pack:
A wall mounted unit in the equipment area which supplies a low dc voltage to the cell. The Power Pack is electrically connected to the pool pump and operates whenever the pump is turned on.
Cell:
The cell consists of a number of titanium mesh plates assembled in a housing which is positioned in the return to pool line after the filter. Pool water flows across these plates.
Electrolyte:
The pool water with a salt content of around 6000ppm (0.6%) forms the electrolyte. An average 60,000 litre pool will initially require in the order of 350kg of salt to reach this level. Salt is not lost by evaporation, however, addition has to be made to account for solution lost to backwash, rain and splash out.
Variable factors governing the quantity of chlorine produced:
Salt Level:
The salt level must be maintained at optimum to ensure full production. Salt levels should be tested with a correctly maintained and calibrated electrical conductivity meter. Some chlorinators incorporate a meter or light system which gives a broad indication as to the salt level.
Cell Cleanliness and Age:
Calcium type deposits form on the cell and impair efficiency. Some units ramp down chlorine production when the salt level drops to below minimum salt level or when water temperature drops below 15C. The cell has a working life of around 3 years and will need to be replaced. As a cell deteriorates with age it may be necessary to increase the salt level to compensate for the drop in production.
Run Period:
If the chlorinator is correctly sized, it will in summer season produce sufficient chlorine in 6-10 hours running. If the salt level is low, the cell dirty, or the cell worn the unit will not operate to optimum and that time will need to be extended to whatever is necessary to produce sufficient chlorine. Both systems are fixed output and cannot account for changing body load. In a domestic situation, upcoming high demand periods may be known and supplementary hand chlorination to raise residual prior to usage is the best solution. It is important to be able to maintain an adequate chlorine residual while swimmers are in the pool. To hand chlorinate after loss of residual and clarity is not an acceptable option.
SERVICE REQUIREMENTS;
Liquid Chlorine:
Injection point to be cleaned monthly (5 mins). Pump tube to replaced every 6 months (5 mins).
Salt Chlorinator:
Cell to be cleaned 6 monthly or monthly for rare examples of non self cleaning cells (15 - 30 mins). Salt level to be maintained monthly or more often in peak period or rain season.
General:
Both systems are superior to hand chlorination. They are to a large degree self maintaining and importantly add chlorine over the filtration cycle.
For domestic pools either system will work satisfactorily if correctly sized to the pool. The support chemistry requirement is the same for both systems. Important
Note:
Chlorine activity or its bacteria killing power is pH dependent. pH must be regularly checked and adjusted as necessary. At pH 8.00, chlorine is 22% effective, at pH 7.2 chlorine is 66% effective. If you have a test sample of 1.0 ppm free chlorine at pH 8.0, in terms of activity this is really equivalent to a chlorine reading of 0.2ppm.

Chlorine dioxide is a powerful oxidising disinfectant used worldwide for water treatment, food sanitation, and industrial processes. It is not the same as chlorine; it behaves very differently in water, and must be handled with strict safety controls due to its reactivity and toxicity at high concentrations.
🧪 What Chlorine Dioxide Is
-
A yellow‑green gas with the formula ClO₂.
-
A strong oxidiser, not a chlorinating agent (unlike chlorine gas or hypochlorite).
-
Highly soluble in water but does not hydrolyse—it remains as dissolved gas.
-
Boiling point: 11 °C, meaning it must be generated on‑site or stored cold.
⚙️ How It Works (Mechanism of Action)
Chlorine dioxide inactivates microorganisms through electron transfer:
-
It oxidizes cell membranes, proteins, and nucleic acids
-
It disrupts biosynthesis and metabolic pathways
-
It is effective against:
-
Bacteria
-
Viruses
-
Protozoa
-
Fungi
-
Biofilms (one of its major strengths)
This broad-spectrum activity is why it’s used in drinking water treatment, cooling towers, food processing, and industrial sanitation
💧 Approved & Legitimate Uses
According to environmental and public health authorities:
✔ Water Treatment
-
Used in municipal drinking water systems at very low concentrations
-
Effective for taste/odour control, iron/manganese oxidation, and biofilm removal
✔ Industrial & Commercial Uses
-
Pulp and paper bleaching (major global use)
-
Food processing sanitation
-
Cooling tower disinfection
-
Dairy equipment sanitation
-
Algaecide, bactericide, and fungicide in swimming pools and spas (in regulated applications)
-
Effective against Giardia, Cryptosporidium, and other chlorine‑resistant organisms.
⚠️ Safety, Toxicity & Misuse Risks
This is where clarity matters.
Toxicity at High Doses
-
High concentrations can damage red blood cells and irritate the GI tract
-
Inhalation of gas can cause respiratory irritation or worse
-
Concentrated solutions are unstable and can explode if mishandled
Misuse in Alternative Health Circles
Some groups promote chlorine dioxide as a “cure” for various diseases.
There is no scientific evidence supporting these uses, and health authorities warn strongly against ingestion of concentrated solutions.


Safety & Health Considerations
-
Safe only at very low concentrations used in regulated water treatment.
-
High doses can damage red blood cells and irritate the GI tract.
-
Not approved for ingestion or unregulated “protocols” marketed online.
-
Requires proper ventilation and chemical handling procedures.
Chemical used



The pH level, or potential of hydrogen, indicates the acidity or alkalinity of pool water, determined by the concentration of hydrogen ions. It is measured on a scale ranging from 0 to 14. As outlined in the Halogen/Chlorine module, maintaining the correct pH is vital for achieving the overall objectives of water chemistry in pools.
PROVIDE WATER THAT IS:
BACTERIA FREE.
ALGAE FREE.
NOT IRRITANT TO SWIMMER EYES, NOSE
AND SKIN.
AESTHETICALLY PLEASING, FREE FROM TASTE AND ODOUR.
THAT IS IN A PASSIVE STATE TO ITS SURROUNDS.
Let's discuss pH in more detail, this may be technical in nature, but this is essential to express how pH is calculated and how it is affected by chemical, and environmental intervention.
What is pH?
pH is a quantitative measure of the acidity or basicity of aqueous or other liquid solutions. It translates the concentration of hydrogen ions into numbers between 0 and 14. In pure water, which is neutral, the pH is 7. A solution with a pH less than 7 is considered acidic, while a pH greater than 7 indicates basic or alkaline properties. Essentially, pH represents the balance between acidic and alkaline components in a solution.
Why is pH Important?
Chlorine effectiveness: Proper pH ensures chlorine works optimally.
Swimmer comfort: Balanced pH prevents skin and eye irritation.
Equipment protection: Maintaining the right pH prevents damage to pool surfaces and plumbing.
Effect of rain on pH levels
Heavy or prolonged rain events can affect the pH levels in swimming pools. While pH levels may appear to rise while it is raining due to aeration of the surface. In the long term rainwater is typically slightly acidic, with a pH around 5.0. When it mixes with your pool water, it can lower the pH, making the water more acidic. This can lead to discomfort for swimmers and potential damage to pool equipment and surfaces.
Additionally, rain can introduce contaminants and dilute the chemicals in your pool, further disrupting the chemical balance. It’s important to test and adjust the pH levels after heavy rainfall to maintain a safe and comfortable swimming environment.
A need to adjust the pH levels after rain is essential?
Effect of aeration on pH levels
Aeration can have a significant impact on the pH level of swimming pool water.
Here’s how it works:
Increases pH:
Aeration introduces air into the water, which increases the rate of carbon dioxide outgassing. This process reduces the amount of carbonic acid in the water, thereby raising the pH level without affecting total alkalinity.
No Chemical Use:
It’s a natural method to adjust pH levels, making it a great option if you prefer to avoid adding chemicals to a pool.
Time Factor:
The time it takes for aeration to raise the pH can vary. Unlike chemical adjustments, which can be relatively quick, aeration might take several days to show noticeable changes.
Additional Benefits:
Besides adjusting pH, aeration can also help cool down pool water, especially in hot climates, and improve overall water circulation.
Forms of aeration:
Air blowers, waterfalls, water blades, fountains, heavy or prolonged rain events.
Key considerations:
Utilizing aeration to adjust pH levels is not a viable method. For accurate pH testing, select a representative sample away from waterfalls, fountains, operational blowers, or during intense or extended rainfall.
For pools or spas equipped with aeration devices, it is crucial to inform customers about the necessity of consistent pH monitoring and adjustment, or the implementation of automatic pH control and dosing systems.
Effects of pH Imbalance:
High pH:Causes calcium scaling on pool surfaces.
Reduces chlorine effectiveness.
Leads to cloudy water.
Low pH:Corrodes pool materials (plaster, metals, grout, tiles).
Increases chlorine effectiveness, but risks water being too corrosive.
Testing and adjusting pH:
Regularly test pH using a reliable kit.
To raise pH (if too low), add buffer (sodium bi-carbonate) or soda ash (sodium carbonate).
To lower pH (if too high), add muriatic acid or dry acid (sodium bisulfate).
pH is defined as the negative logarithm of the concentration of H+ ions. As a result, the meaning of the name is justified as hydrogen power.
We know that not all acids and bases react at the same rate with the same chemical compound. Some react violently, others moderately, and still others do not. To quantify the strength of acids and bases, we use a universal indicator that changes color depending on the concentration of hydrogen ions in the solution. In general, the value of acids and bases is used to quantify their strength.
Although less intuitive, the mathematical definition is more useful overall. It states that pH equals the negative logarithmic value of hydrogen ion (H+) concentration.
pH = -log [H+]
The pH level is determined by the activity of hydrogen atoms, which is a good indicator of the acidity or alkalinity of water. The scale ranges from 0 to 14, with 7.0 being neutral. Water with a low pH is said to be acidic, while water with a high pH is said to be basic, or alkaline.
Acids and Bases Review
There are several ways to define acids and bases, but pH specifically refers to the concentration of hydrogen ions in aqueous (water-based) solutions. Water dissociates into a hydrogen ion and a hydroxide.
Water shows amphoteric nature (it can act as both an acid and a base). Two water molecules react to produce hydronium and hydroxide ions:
This is also called the self-ionisation of water.
This equation can also be represented as;
The reaction can be shifted to the reactants or products, as expected for any equilibrium:
When an acid (H+) is added to water, the equilibrium shifts to the left, and the concentration of OH– ions decreases.
When a base (OH–) is added to water, the equilibrium shifts to the left, and the concentration of H+ decreases.
When calculating pH, keep in mind that [ ] stands for molarity, M. Molarity is measured in moles of solute per litre of solution. If the concentration is given in a unit other than moles (mass percent, molality, etc.), convert it to molarity before applying the pH formula.
he relationship between pH and molarity can be expressed as:
Kw = [H+] [OH–] = 1 x 10-14 at 25°C
for pure water, [H+] = [OH–] = 1 x 10-7
Kw is the dissociation constant of water
Acidic Solution: [H+] > 1×10-7
Basic Solution: [H+] < 1×10-7
The pH of Acids and Bases
A solution’s pH ranges from 0 to 14.
In conclusion:
Due to the role that pH plays in the efficiency of chlorine and in the effects it can have on pool surface, equipment and reliability of chemical test results.
Constant monitoring and adjustment of pH is crucial, we will provide parameters and dose rates in another module. We will discuss how Total Alkalinity acts as a pH stabiliser and is an important component in water chemistry is next.
If you are interested in learning more about this subject, go to the following online resources for a deeper dive.
pH - Definition, Calculation, and Significance (scienceinfo.com)
What Is pH? The pH Formula & Equation | ChemTalk (chemistrytalk.org)
pH Definition and Equation in Chemistry (thoughtco.com)
Chemicals Related to reducing pH

Hydrochloric Acid, also known as Muriatic acid. Normally with a 32% concentration

Also Known as Dry Acid, Sodium bisulphate monohydrate.
Hydrochloric Acid Vs Sodium Bisulphate
Hydrochloric Acid (Muriatic Acid):
Pros: Highly Effective: HCl is a potent acid that effectively lowers both pH and total alkalinity in pool water.
Stain and Scale
Removal: It can remove mineral deposits, stains, and scale buildup from pool surfaces.
Quick Action: HCl works rapidly to adjust water chemistry.
Cons:
Safety Hazard: Handling HCl requires caution due to its corrosive nature.
It can cause burns and respiratory irritation.
Storage and Handling:
Proper storage and protective gear are essential.
Cost:
HCl is generally more affordable than sodium bisulfate. Environmental Impact: Disposal of HCl must be done carefully to avoid harm to the environment.
Sodium Bisulphate (Dry Acid):
Pros:
Safer to Handle:
Dry acid (sodium bisulphate) is granulated and less hazardous than HCl.
Ease of Use:
It dissolves easily in water, making it convenient for pH adjustment.
Algae Prevention: Helps prevent algae growth by maintaining proper pH.
Compatibility: Compatible with various pool types and other chemicals.
Cons:
Cost:
Sodium bisulfate is more expensive than HCl.
Slower Action: It may take longer to adjust pH compared to HCl.
Environmental Impact:
While safer, it still produces an acidic solution when mixed with moisture.
Choosing the Right Acid: Safety First:
If safety is a top priority, opt for sodium bisulphate.
Efficiency: If rapid pH adjustment is crucial, consider HCl. Budget: Consider cost implications.
Long-Term:
Evaluate long-term costs and environmental impact.
Understanding Total Alkalinity in Water Chemistry
Total Alkalinity (TA) is a key parameter in water chemistry—especially for pools, spas, and natural aquatic systems—because it acts as a buffer to stabilize pH levels. Here's a deeper dive into how it works and why it matters:
What Is Total Alkalinity?
-
Definition: TA is the measure of water's ability to neutralize acids, primarily due to the presence of bicarbonate, carbonate, and hydroxide ions.
-
Units: Measured in parts per million (ppm) as calcium carbonate (CaCO₃).
-
Target Range (in pools): Typically between 80–120 ppm, depending on the sanitizer used and surface type.
The Role of TA in pH Stability
-
Acts as a buffer against pH fluctuations.
-
High TA can make pH resistant to downward shifts—but also harder to adjust.
-
Low TA makes the pH prone to rapid changes, risking corrosion or swimmer discomfort.
Think of TA like a shock absorber for your water’s pH—too little, and every chemical you add hits hard; too much, and adjustments become sluggish.
Interactions with Other Parameters
Parameter Effect of TA
pH Stabilizes swings, but extreme levels alter control
Calcium Hardness Indirect influence due to precipitation dynamics
Sanitizer Efficiency Low TA/pH may increase sanitizer activity, but also corrosion risk
Scaling/Corrosion Low TA → corrosion; High TA → scale formation
Adjusting TA
-
To raise TA: Add sodium bicarbonate (baking soda).
-
To lower TA: Use acid treatments (like muriatic acid or sodium bisulphate), often combined with aeration to isolate TA adjustments without plummeting pH.
Real-World Implications (like your pool assessments)
In aged pools with scaling or staining:
-
High TA can promote calcium carbonate deposition, worsening scale.
-
Low TA risks pH volatility, which can accelerate surface damage or stain development.
Balancing TA becomes essential not only for maintaining water clarity and safety but also for prolonging pool surface integrity—especially critical if the property’s up for sale.
Adjusting pH and Total Alkalinity
-
To Raise Total Alkalinity
-
Add: Sodium bicarbonate (baking soda)
-
Effect: Raises TA and slightly increases pH
-
Dosage: ~1 kg per 50,000 L to raise TA by 10 ppm
-
To Lower Total Alkalinity
-
Add: Muriatic acid or sodium bisulphate
-
Method: Add slowly with pump running; circulate for 4–6 hours
-
Effect: Lowers both TA and pH — so monitor carefully
-
To Raise pH (without affecting TA much)
-
Add: Sodium carbonate (soda ash)
-
Effect: Raises pH more than TA
-
Use Case: When pH is low but TA is in range
-
To Lower pH
-
Add: Muriatic acid or sodium bisulfate
-
Effect: Lowers both pH and TA
-
Tip: Use in small doses to avoid overshooting
-
Pro Balancing Strategy
-
Test first: Use a reliable test kit or digital meter.
-
Adjust TA first: It buffers pH, so get it right before fine-tuning pH.
-
Wait & retest: After chemical addition, wait 6–24 hours before retesting.
-
Avoid chasing numbers: Aim for stability, not perfection.
-
How Total Alkalinity Influences pH
-
Buffering Effect: TA acts as a stabilizer, absorbing acids or bases to prevent sudden pH changes. Think of it as a shock absorber for your pool’s pH.
-
High TA: Makes pH resistant to change — but if too high, it can cause pH to drift upward over time.
-
Low TA: Leaves pH vulnerable to wild swings, especially after adding chlorine or acid.
-
Ideal Balance Strategy
Parameter Ideal Range Role in Stability
Total Alkalinity 80–120 ppm Buffers pH against rapid changes
pH 7.4–7.6 Ensures swimmer comfort & chlorine efficacy
-
Adjusting one affects the other. For example:
-
Adding sodium bicarbonate raises TA and slightly nudges pH upward.
-
Adding acid lowers both TA and pH — so you need to dose carefully.
-
-
Pro Insight:
The Effect of Total Alkalinity on Swimming Pool Water Quality
1. pH Stability (Primary Effect)
Total Alkalinity is the buffer that helps keep pH from changing too quickly.
When TA is correct, pH changes slowly and predictably.
-
Low TA → pH becomes unstable (“pH bounce”)
-
High TA → pH drifts upward and becomes difficult to control
This is the single most important operational effect of TA.
2. Chlorine Efficiency
Because chlorine works best in a stable pH range, TA indirectly affects sanitiser performance.
-
Low TA: pH swings reduce chlorine efficiency
-
High TA: pH rises, reducing the proportion of active hypochlorous acid.
Result: chlorine becomes less effective at killing pathogens and oxidising contaminants.
3. Water Balance & LSI
-
TA is a major component of the Langelier Saturation Index (LSI).
-
Low TA → corrosive water
-
Etching of plaster
-
Corrosion of metal components
-
Premature equipment wear
-
High TA → scale-forming water
-
Calcium scale on tiles, heaters, salt cells
-
Cloudy water
-
Reduced heat transfer efficiency.
4. Swimmer Comfort
Stable pH (supported by correct TA) reduces:
-
Eye and skin irritation
-
Strong chlorine odours (caused by poor pH control and chloramine formation)
5. Chemical Consumption
Incorrect TA increases chemical usage:
-
Low TA: more acid and base required to chase pH
-
High TA: more acid required to bring pH down, often repeatedly
Correct TA reduces operational cost and stabilises the system.
The Effect of Total Alkalinity on Swimming Pool Equipment
1. Corrosion Risk (When TA Is Too Low)
Low TA makes the water aggressive because pH becomes unstable and often drops. This leads to:
-
Corrosion of metal components (ladders, handrails, screws, light rings)
-
Damage to pump seals and heater internals
-
Etching of plaster, grout, and tile surfaces
-
Shortened lifespan of salt cells due to acidic conditions
Low TA essentially removes the protective buffering that keeps water from attacking surfaces and metals.
2. Scaling Risk (When TA Is Too High)
High TA pushes pH upward, creating scale‑forming conditions. This causes:
-
Calcium scale inside heaters, reducing heat transfer
-
Scale on salt chlorinator plates, reducing chlorine output
-
Deposits on tiles, skimmer throats, and waterline surfaces
-
Increased pump strain due to restricted flow in pipes and fittings
High TA makes the water “over‑buffered,” encouraging carbonate precipitation.
3. Increased Chemical Load on Equipment
Incorrect TA forces the system to work harder:
-
More acid dosing → more wear on dosing pumps and injectors
-
More pH correction cycles → more stress on automation systems
-
More chlorine demand → higher load on salt cells and feeders
Stable TA reduces the operational burden on all equipment.
4. Reduced Chlorine Efficiency → More Wear
When the TA is out of range, pH becomes unstable or drifts high. This reduces chlorine efficiency, which leads to:
-
More contaminants are reaching the filters
-
More oxidation load on heaters and metal components
-
More strain on sanitising equipment
Poor TA indirectly increases equipment workload.
-
TA Interplay with Sanitisers
-
Chlorine: TA stabilises pH, ensuring chlorine remains effective.
-
Bromine: Works best in slightly higher TA ranges (100–150 ppm).
-
Saltwater Systems: TA helps buffer pH fluctuations caused by electrolysis.
-
Ozone & UV: TA indirectly supports these systems by maintaining stable pH levels, which optimise their efficacy.
-
Sodium Bicarbonate vs. Soda Ash
-
Sodium Bicarbonate (Baking Soda): Primarily used to raise TA without significantly affecting pH.
-
Soda Ash (Sodium Carbonate): Used to raise pH more dramatically, with a smaller impact on TA.
-
Key Differences:
-
Sodium bicarbonate is gentler and ideal for fine-tuning TA.
-
Soda ash is more aggressive and suited for rapid pH adjustments.
-
-
Usage Tips:
-
Test water parameters before adding either chemical.
-
Add gradually and retest to avoid overshooting desired levels.
-
 |  |
|---|---|
 |  |
Chemicals Related to the control of Alkalinity
Calcium Hardness in Pool Chemistry
Why It Matters:
Calcium hardness refers to the concentration of dissolved calcium ions in your pool water. It’s a key component of water balance and plays a crucial role in protecting your pool surfaces and equipment.
Why It’s Important Surface Protection:
Damage to Pool Surface:
Low calcium levels make water aggressive, causing it to leach calcium from concrete, plaster, or fibreglass surfaces—leading to etching or gelcoat damage.
Scale Prevention:
High calcium levels can cause scaling on surfaces and inside pipes, especially when combined with high pH. and T.A. Water
Clarity & Balance: Imbalanced calcium hardness can lead to cloudy water and interfere with pH and alkalinity control.
Ideal Levels Recommended Range:
200–400 ppm (parts per million) is generally ideal.
Below 200 ppm: Risk of corrosion and surface damage. Above 400 ppm: Increased risk of scaling and cloudy water.
Testing Calcium Hardness:
Test kits use liquid reagent kits for calcium-specific readings or test strips for total hardness (includes magnesium).Frequency: Every few months is sufficient, as calcium levels are relatively stable.
Adjusting Calcium Hardness:
-
To increase: Add calcium chloride or similar products.
-
To Lower: Partially drain and refill with low-calcium water (e.g., rainwater or tap water, depending on your region).
Note: Chemical “reducers” only bind calcium temporarily—they don’t remove it permanently.
Pool Type Considerations Fibreglass Pools:
Require calcium 175ppm to 225ppm to protect the gelcoat. Zero calcium can void warranties.
Pool Type Considerations Vinyl Lined Pools:
Require calcium 150ppm to 250ppm to protect the integrity of vinyl. Zero calcium can void warranties.
Concrete Pools:
Naturally release calcium over time, especially when new. When managing a pool on the Sunshine Coast, local water sources and climate might influence your calcium levels range of 200ppm to 400ppm should be your target.
Total Hardness
Total Hardness is a vital—but often misunderstood—parameter in swimming pool chemistry.
It's not just a measure of calcium; it encompasses all dissolved polyvalent metal ions, with calcium and magnesium being the major players.
Here’s a structured overview to complement the detail already present on your page:
What Is Total Hardness?
Total Hardness = Calcium Hardness + Magnesium Hardness (primarily). Measured in parts per million (ppm) using test kits or strips Ideal range for pools: 200–400 ppm.
Why It Matters Low Total Hardness (<150 ppm):
Water becomes corrosive Can damage plaster, pool equipment, and metal components. May cause etching on pool surfaces
High Total Hardness (>500 ppm):
Encourages scale formation (especially on heaters and tiles). Affects clarity and may cause cloudy water. Interacts with other parameters like pH and alkalinity
Adjustment Strategies
To Increase Hardness:
Add calcium chloride (especially in soft water regions). Retest after 24 hours to ensure balanced uptake
To Decrease Hardness:
Partially drain and dilute with fresh water. Use reverse osmosis systems (more advanced)
Interplay with Other Parameters. Total Hardness interlocks tightly with pH, Total Alkalinity, and Calcium Saturation Index (CSI). When magnesium levels are elevated, CSI calculations can become skewed, potentially masking aggressive or scaling water tendencies.
Understanding the Relationship Total Hardness vs. Calcium Hardness:
Total hardness includes calcium and magnesium ions. Calcium hardness measures only calcium ions. Potassium doesn’t directly contribute to hardness but may influence water balance through other chemical interactions.
Magnesium's Role:
Magnesium contributes to total hardness and can compete with calcium in scaling reactions. High magnesium levels may reduce calcium’s tendency to precipitate, slightly mitigating scale formation.
Potassium's Role:
Potassium is typically introduced via potassium-based sanitisers or oxidisers. It doesn’t affect hardness directly but can influence ionic strength and water balance, which may subtly shift calcium solubility—practical Implications for Pool Chemistry. If you're using mineral systems (such as magnesium-based sanitisers), your test strips may show elevated total hardness even if the calcium level is within range. This can lead to misinterpretation of water balance if you're not distinguishing between calcium and total hardness.
Magnesium-rich pools may feel “softer” and be gentler on skin, but they still require calcium to protect surfaces—especially in fibreglass pools.
Chemicals Related to Controlling Calcium Hardness
 |  |  |
|---|---|---|
 |  |  |
 |

The Cyanuric Acid / Stabiliser Story
Summary
Cyanuric Acid is an essential UV‑shielding agent for outdoor pools, but its benefits are tightly constrained by Australian standards and health guidelines. Maintaining stabiliser within the recommended range ensures:
-
Effective chlorine performance
-
Compliance with regulatory requirements
-
Predictable sanitisation outcomes
-
Reduced operational risk
Excess stabiliser compromises disinfection and can only be corrected through water replacement, making proactive monitoring critical.
— Sunlight vs Chlorine
Outdoor pools lose chlorine rapidly under UV exposure. In full sun, most free chlorine can be destroyed within an hour if unprotected.
— Enter Cyanuric Acid (Stabiliser)
Cyanuric Acid binds with free chlorine to form a protective buffer. This slows UV degradation and stabilises chlorine levels throughout the day.
— The Chemistry Link
CYA forms weak, reversible bonds with hypochlorous acid (HOCl). This reduces the unbound HOCl fraction but preserves enough active chlorine for effective disinfection when kept within recommended limits.
— Standards‑Aligned Operating Range
Australian guidance (AS 3633, state health codes, Qld Health) supports maintaining stabiliser at 30–50 ppm for outdoor pools. Upper limits typically sit at ≤ 100 ppm to avoid chlorine suppression.
— When CYA Is Too Low
Low stabiliser (< 20–30 ppm) leaves chlorine unprotected. UV burn‑off increases, residuals drop quickly, and chlorine consumption rises.
— When CYA Is Too High
Excess stabiliser (> 60–100 ppm depending on jurisdiction) reduces HOCl availability, slows pathogen kill times, lowers ORP, and impairs shock treatments.
— How CYA Enters the System
Stabiliser is added intentionally (granular CYA) or unintentionally through stabilised chlorines (dichlor, trichlor). Continuous use of stabilised products causes gradual CYA accumulation.
— Monitoring Requirements
Routine testing is required under AS 3633 and state health guidelines. Turbidimetric or photometric methods ensure accurate readings, especially when stabilised chlorine is used.
— Corrective Action Pathway
There is no approved chemical method in Australia for reducing CYA in situ. The only reliable correction is partial drain and refill, following local discharge regulations.
— Adjusting Operations When CYA Is Elevated
If CYA is high but still compliant, operators may need to increase free chlorine targets, adjust ORP setpoints, or enhance oxidation strategies (UV/ozone) to maintain disinfection performance.
— The Balanced Outcome
Correctly managed stabiliser protects chlorine, maintains sanitising efficiency, and ensures compliance with Australian standards — without compromising water clarity or safety.
— The Core Principle
Cyanuric Acid is the pool’s sunscreen: essential in the right amount, problematic when over‑applied. Balance is the key to reliable, standards‑compliant disinfection.
🏊♂️ LSI: The Story of What Your Water Wants to Do
🧱 1. Your pool is made of calcium
Your pool surface, grout, tile, and even parts of your equipment all contain calcium‑based materials.
This is the “stuff” the water interacts with every day.
💧 2. Water always leans one way or the other
Water is never neutral on its own — it’s always trying to dissolve calcium or drop excess calcium as scale.
LSI simply tells us which direction it’s leaning.
📊 3. LSI is the score
LSI is the water’s “balance score.”
It combines pH, alkalinity, calcium hardness, and temperature into one number that predicts the water’s behaviour.
⚖️ 4. We aim for zero
When LSI is around 0.0, the water is “at peace” with your pool — not dissolving calcium, not depositing it.
Just stable, predictable, and protective.
Langelier Saturation Index in swimming pools
You’re asking a “master variable” question—LSI is where pool chemistry stops being a list of numbers and becomes a system. Let’s unpack it properly.
What the Langelier Saturation Index actually is
The Langelier Saturation Index (LSI) is a calculated number that tells you whether water is:
-
Undersaturated with calcium carbonate → corrosive/etching
-
In equilibrium with calcium carbonate → “balanced”
-
Oversaturated with calcium carbonate → scale-forming
In pool language: it predicts whether your water will dissolve calcium (from plaster, grout, tile, heaters) or deposit calcium scale (on surfaces, heaters, salt cells, tiles).
LSI is not a chemical you add; it’s a way of combining several parameters into one stability number.
The core idea: calcium carbonate equilibrium
Pool surfaces (plaster, grout, some tiles, and concrete) and many components (heat exchangers and salt cells) are affected by calcium carbonate balance.
-
If water is “hungry” for calcium carbonate (LSI negative), it will pull it from surfaces—etching, pitting, roughness, and metal corrosion.
-
If water is “overfed” with calcium carbonate (LSI positive), it will dump the excess as scale—white crust, cloudy water, and heater inefficiency.
LSI quantifies how far your water is from that equilibrium point.
The LSI formula for pools
The classic pool-industry form of LSI is:
[ \text{LSI} = \text{pH} + \text{CF} + \text{AF} + \text{TF} - 12.1 ]
Where:
-
pH = measured pool pH
-
CF = Calcium factor (from calcium hardness)
-
AF = Alkalinity factor (from carbonate alkalinity)
-
TF = Temperature factor (from water temperature)
12.1 = constant representing TDS and standard conditions (approximate)
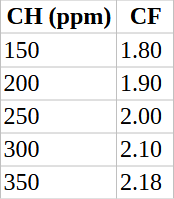
1. Calcium factor (CF)
Based on calcium hardness (CH) in ppm as CaCO₃:
-
CH 75 → CF ≈ 1.5
-
CH 150 → CF ≈ 1.8
-
CH 200 → CF ≈ 1.9
-
CH 300 → CF ≈ 2.1
-
CH 400 → CF ≈ 2.2
-
CH 800 → CF ≈ 2.5
(Exact values come from log-based tables; most pool charts give a quick lookup.)
2. Alkalinity factor (AF)
Based on carbonate alkalinity, not just “total alkalinity” on the test strip.
If you have cyanuric acid (CYA) or borates, you should subtract their contribution to get carbonate alkalinity. Many simplified pool guides still use total alkalinity directly, but strictly speaking:
[ \text{Carbonate alkalinity} \approx \text{Total alkalinity} - \text{CYA correction} ]
Then use a table:
-
Alk 50 → AF ≈ 1.4
-
Alk 100 → AF ≈ 2.0
-
Alk 150 → AF ≈ 2.2
-
Alk 200 → AF ≈ 2.3
Again, charts give the exact factor.
3. Temperature factor (TF)
Based on water temperature:
-
10 °C → TF ≈ 0.3
-
15 °C → TF ≈ 0.4
-
20 °C → TF ≈ 0.5
-
25 °C → TF ≈ 0.6
-
30 °C → TF ≈ 0.7
-
35 °C → TF ≈ 0.8
Warmer water increases TF, pushing LSI upward (more scale-forming tendency).
4. The constant 12.1
This is a composite constant that assumes:
-
Typical TDS (around 1000 ppm)
-
Standard atmospheric conditions
At very high TDS (e.g., older salt pools), some methods adjust this constant slightly, but 12.1 is the standard pool-industry value. •
Salt does not directly change LSI as pH or calcium hardness do.
• But by raising TDS, it nudges LSI downward.
• In saltwater systems, you often need:
• Slightly higher calcium hardness
• Slightly higher alkalinity
• Careful pH management
to keep LSI in the ideal range (around –0.3 to +0.3, depending on your pool surface and climate).
Interpreting LSI values in pools
Typical interpretation:
-
LSI < -0.3 → Aggressive/corrosive water (etching risk)
-
LSI between -0.3 and +0.3 → Acceptably balanced
-
LSI > +0.3 → Scale-forming water
Many professionals aim tighter, around -0.1 to +0.1, especially for delicate finishes or expensive equipment.
What low LSI does (too negative)
-
Etching of plaster and grout → rough, pitted surfaces
-
Metal corrosion → heaters, ladders, light niches
-
Increased dissolved metals → staining later when conditions change
-
Shortened life of concrete/plaster finishes.
-
What high LSI does (too positive)
-
Calcium scale on tiles, waterline, and inside pipes
-
Heater scaling → poor heat transfer, higher energy use, possible failure
-
Salt cell scaling → reduced chlorine production, more cleaning
Cloudy water from precipitated calcium carbonate.
Why LSI matters more than “just pH and alkalinity”
Most basic pool advice says: “Keep pH, alkalinity, and calcium hardness in range.” The problem is that each parameter interacts:
-
High pH + high CH + warm water can be scale-forming even if alkalinity is “in range”.
-
Low pH + low CH can be corrosive even if alkalinity looks okay.
LSI integrates these into one stability number, so you see the overall tendency instead of chasing individual readings.
A worked example for a typical pool
Imagine a residential pool with:
-
pH = 7.6
-
Calcium hardness = 250 ppm
-
Total alkalinity = 90 ppm (assume low CYA for simplicity)
-
Temperature = 26 °C
Using typical factor tables:
-
CH 250 → CF ≈ 2.0
-
Alk 90 → AF ≈ 1.9
-
Temp 26 °C → TF ≈ 0.6
Now calculate:
[ \text{LSI} = 7.6 + 2.0 + 1.9 + 0.6 - 12.1 ]
[ \text{LSI} = 12.1 - 12.1 = 0.0 ]
So this water is right at equilibrium—neither strongly scale-forming nor corrosive.
If the same pool warms to 32 °C (TF ≈ 0.75) without any other changes:
[ \text{LSI} = 7.6 + 2.0 + 1.9 + 0.75 - 12.1 = 0.15 ]
Now it’s mildly scale-forming—you might start to see more tendency for scale on heaters and salt cells.
How to adjust water using LSI
Think of LSI as a “target” and each parameter as a lever you can move.
1. If LSI is too low (corrosive)
You can:
-
Raise pH (within safe range, e.g., toward 7.6–7.8)
-
Increase calcium hardness (add calcium chloride)
-
Increase alkalinity (add sodium bicarbonate)
-
Allow temperature to rise (not always practical, but matters in heated pools)
In plaster pools, raising calcium hardness is often the most protective move, especially for new or freshly resurfaced pools.
2. If LSI is too high (scale-forming)
You can:
-
Lower pH (toward 7.2–7.4)
-
Lower alkalinity (acid + aeration method)
-
Avoid unnecessary calcium additions
-
Manage temperature (e.g., not overheating spas more than needed)
In saltwater pools, controlling pH and alkalinity is usually the most effective way to keep LSI in check and protect the cell.
Special considerations in real pools
Cyanuric acid (CYA)
Stabiliser (CYA) ties up some alkalinity. For a more accurate LSI:
-
Correct alkalinity by subtracting a portion of CYA from total alkalinity.
-
This gives carbonate alkalinity, which is what LSI really uses. Salt Water Pool and Spa
Many field calculations skip this step and still get “good enough” results, but for tight control—especially in commercial or high-value pools—it matters.
Saltwater pools and high TDS
Salt pools have higher TDS, which slightly shifts the equilibrium. Some advanced LSI methods adjust the constant (12.1) or add a TDS factor. For most residential pools, the standard 12.1 constant still gives a useful, practical LSI.
Surface type
-
Plaster/quartz/pebble: LSI is critical—these surfaces are calcium-based.
-
Vinyl/fibreglass: Less vulnerable to etching, but heaters, metal parts, and salt cells still care about LSI.
So even in non-plaster pools, LSI is still relevant for equipment protection.
Limitations and common misconceptions
1. LSI is a model, not a guarantee.
It predicts tendency, not exact outcomes. Local conditions, micro-environments (inside heaters, salt cells), and water movement can create hot spots of scaling or corrosion even when bulk LSI looks okay.
2. “Balanced LSI” doesn’t mean “sanitised.”
You can have perfect LSI and still have unsafe water if chlorine, ORP, or filtration are poor. LSI is about water balance, not disinfection.
3. You don’t have to chase a single “perfect” number.
A range around zero (e.g., -0.1 to +0.1) is realistic. Trying to nail exactly 0.00 will drive you mad and isn’t necessary.
4. Test accuracy matters.
Because LSI is a calculation, errors in pH, CH, or alkalinity propagate into the result. Good testing (fresh reagents, proper technique) is essential.
How to teach LSI simply
For educational signage, training, or client explanations, a simple narrative works well:
-
“Your pool surface and equipment are made of calcium-based materials.”
-
“Water either wants to dissolve calcium or dump extra calcium as scale.”
-
“LSI is the score that tells us which way your water is leaning.”
-
“We aim for around zero so the water is ‘at peace’ with your pool.”
Here’s a clean, signage‑ready LSI cheat chart tuned to the parameter ranges you actually use in real pools on the Sunshine Coast — simple enough for clients, accurate enough for technicians, and visually aligned with your schematic‑first style.
Balanced water target: LSI between –0.1 and +0.1
This chart lets you estimate LSI quickly without tables or calculators. It’s built around the ranges you routinely work in: pH 7.2–7.8, TA 60–120, CH 150–350, and 20–32 °C water.
1. Temperature Factor (TF)
2. Calcium Factor (CF)
3. Alkalinity Factor (AF)
(Assumes typical CYA correction already applied — or low CYA pools)
4. Quick LSI Formula
[ \text{LSI} = \text{pH} + \text{CF} + \text{AF} + \text{TF} - 12.1 ]
5. Rapid‑Use LSI Grid (Your Field Sweet Spot)
Find your pH row → add CF → add AF → add TF → subtract 12.1
This grid shows the starting point before adding hardness, alkalinity, and temperature factors.
pH
Base Value (pH – 12.1)
Then simply add:
-
CF from the CH table
-
AF from the alkalinity table
-
TF from the temperature table
If the final number is:
-
< –0.1 → water is etching
-
–0.1 to +0.1 → balanced
-
> +0.1 → water is scaling
6. Three Example “Field Profiles”
These help techs instantly recognise patterns.
A. Typical Sunshine Coast Pool (balanced)
-
pH 7.6 → –4.5
-
CH 250 → CF 2.00
-
TA 80 → AF 1.85
-
Temp 26 °C → TF 0.62
LSI = –4.5 + 2.00 + 1.85 + 0.62 = 0.0
→ Perfectly balanced.
B. Salt Pool in Summer (scale‑prone)
-
pH 7.8 → –4.3
-
CH 300 → CF 2.10
-
TA 100 → AF 2.00
-
Temp 30 °C → TF 0.70
LSI = –4.3 + 2.10 + 2.00 + 0.70 = +0.50
→ High scale risk (salt cell + heater)
C. Fresh Plaster Pool (etch‑prone)
-
pH 7.2 → –4.9
-
CH 150 → CF 1.80
-
TA 60 → AF 1.70
-
Temp 24 °C → TF 0.58
LSI = –4.9 + 1.80 + 1.70 + 0.58 = –0.82
→ Strongly corrosive — raise CH and pH